AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Nutrfs12/11/2023  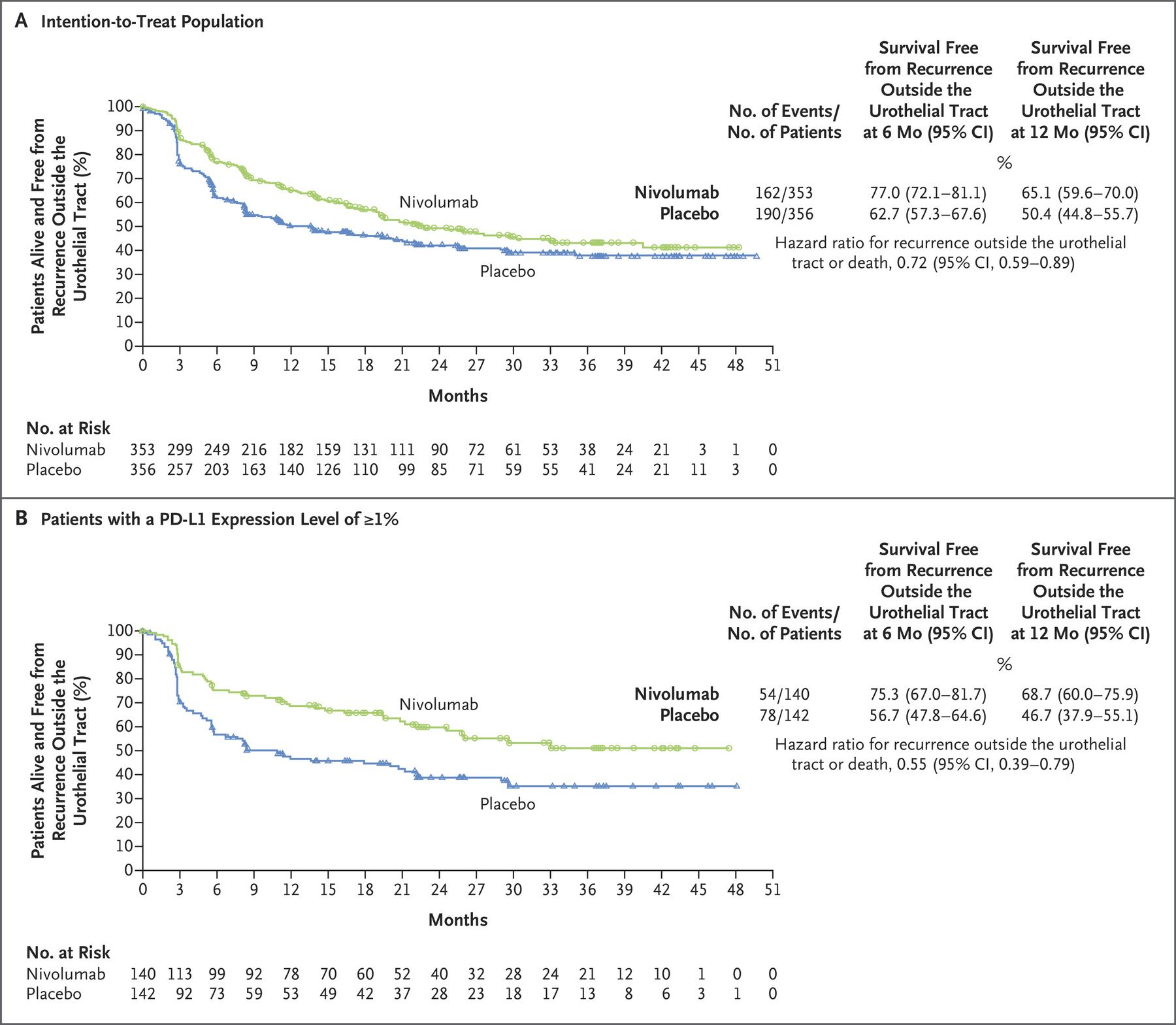
Patients were allowed to receive chemotherapy in the neoadjuvant setting to shrink their tumors before surgical resection. The CheckMate-274 trial randomized approximately 700 surgically resected, high-risk muscle-invasive urothelial carcinoma patients to receive either nivolumab or placebo in the adjuvant setting a subset of patients expressed PD-L1 in more than 1 percent of tumor cells. Currently, the checkpoint inhibitor is approved in the US for locally advanced or metastatic urothelial cancer patients who have progressed on platinum chemotherapy or within a year of neoadjuvant or adjuvant treatment with platinum-containing chemotherapy. Such an approval would allow BMS to market the drug to patients with earlier-stage disease. The company said it will discuss this data with regulators, indicating it is planning to seek marketing approval for nivolumab as an adjuvant treatment for bladder cancer. 
Clinical trial information: NCT02632409.ĬI, confidence interval NE, not estimable, NR, not reached.NEW YORK – Bristol Myers Squibb said on Tuesday that in the Phase III CheckMate-274 trial, adjuvant treatment with nivolumab (Opdivo) nearly doubled disease-free survival in high-risk muscle-invasive urothelial cancer patients compared to placebo, and had an even greater benefit in patients with PD-L1-positive tumors. These results further support adjuvant NIVO as a standard of care for high-risk MIUC after radical resection. The hazard ratio (HR) for DFS and NUTRFS in PD-L1 ≥ 1% patients and for DMFS in both ITT and PD-L1 ≥ 1% patients also continued to improve versus the primary analysis. Conclusions: With extended follow-up, NIVO continued to show DFS, NUTRFS, and DMFS benefits versus PBO. Overall survival will be assessed at a future database lock. Grade 3–4 treatment-related adverse events occurred in 18.2% and 7.2% of patients in the NIVO and PBO arms, consistent with the primary analysis. NUTRFS and DMFS benefits with NIVO versus PBO were also observed in both populations (Table). DFS benefit was seen in most subgroups analyzed including age, sex, ECOG PS, nodal status, prior cisplatin-based chemotherapy, and PD-L1 status. With median follow-up of 36.1 months (minimum follow-up, 31.6 months), median DFS was 22.0 months with NIVO versus 10.9 months with PBO in ITT patients and 52.6 months with NIVO versus 8.4 months with PBO in patients with PD-L1 ≥ 1% (Table). Results: There were 353 patients randomly assigned to NIVO (PD-L1 ≥ 1%, n = 140) and 356 to PBO (PD-L1 ≥ 1%, n = 142). Distant metastasis-free survival (DMFS) and safety were exploratory endpoints. Overall survival and non–urothelial tract recurrence-free survival (NUTRFS) in ITT patients and in patients with PD-L1 ≥ 1% were secondary endpoints. DFS was also analyzed in prespecified subgroups. Primary endpoints were DFS in ITT patients and in patients with PD-L1 ≥ 1%. Patients had pathologic evidence of UC at high risk of recurrence and Eastern Cooperative Oncology Group performance status (ECOG PS) ≤ 1. Patients were randomly assigned 1:1 to NIVO 240 mg every 2 wk or PBO for ≤ 1 year of treatment. Methods: CheckMate 274 is a phase 3, double-blind trial of adjuvant NIVO versus PBO for high-risk muscle-invasive urothelial carcinoma (MIUC) (bladder, ureter, or renal pelvis) after radical resection. Background: The 2 primary endpoints of the CheckMate 274 trial were met as nivolumab (NIVO) improved disease-free survival (DFS) versus placebo (PBO) in the intent-to-treat (ITT) population and in patients with tumor programmed death ligand 1 (PD-L1) expression ≥ 1%. 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
